Anemia in children with chronic kidney disease
Article information
Abstract
Chronic kidney disease (CKD) causes numerous changes that destabilize homeostasis, of which anemia is one of its important complications. Anemia significantly reduces the quality of life in children with CKD and plays a crucial role in the progression of cardiovascular disease such as left ventricular hypertrophy, a major cause of mortality in those with advanced CKD. The treatment of anemia is a pivotal factor in reducing morbidity and mortality rates in children with CKD, representing one of the methods for enhancing patients’ quality of life.
Introduction
Chronic kidney disease (CKD) can lead to various changes that disrupt homeostasis, with anemia as its important complication. Anemia in kidney failure was recognized in 1836 by Richard Bright, an English physician and early pioneer in kidney disease research, who noted that “healthy faces fade over time” [1]. Likewise, in 1839, toxicologist Robert Christison said, “By far the most remarkable character of the blood in the advanced stage of the Bright’s disease is a gradual and rapid reduction of its coloring matter or hematosin” [2]. As kidney disease progresses, anemia prevalence increases [3]. According to the North American Pediatric Renal Trials and Collaborative Studies (NAPRTCS), the prevalence of anemia is defined as 73%, 87%, and over 93% in pediatric CKD stages 3, 4, and 5, respectively [4]. Anemia is a significant factor in reducing the quality of life in children suffering from CKD, and it plays a crucial role in the progression of cardiovascular disease such as left ventricular hypertrophy, a leading cause of mortality among children with advanced CKD [5-9]. In addition, anemia in pediatric CKD significantly increases the risk of hospitalization and mortality compared with no anemia [10-12]. Therefore, this review aims to enhance our understanding of anemia in pediatric CKD and provide a comprehensive overview of its appropriate treatment.
Pathogenesis of anemia in CKD
Since the 1950s, with the kidney being recognized as a key regulator of erythropoiesis and the primary source of erythropoietin (EPO) production, EPO deficiency has emerged as the major probable cause of anemia in CKD [13,14]. CKD-related anemia is a multifactorial process resulting from relative EPO deficiency, uremia-induced inhibition of erythropoiesis, shortened erythrocyte survival, and disordered iron homeostasis [15]. Additionally, factors such as inflammation, infection, blood loss, hyperparathyroidism, and nutritional deficiency play a role in causing anemia in CKD (Table 1) [15-18]. Recent studies have identified that the accumulation of iron-regulatory protein hepcidin, which was upregulated in CKD, is a main contributor to disordered iron homeostasis and anemia in CKD by impairing dietary iron absorption from the gastrointestinal tract and iron mobilization from body stores [15,19,20]. As glomerular filtration rate and inflammation decrease, hepcidin levels are expected to rise. Thus, hepcidin may be a potentially modifiable medium for anemia in CKD, and treatment strategies targeting hepcidin are currently being studied [21-23]. Therefore, evaluating these multiple factors and establishing an appropriate treatment plan are necessary to successfully treat anemia in CKD.
Defining anemia in CKD
Defining and assessing anemia in pediatric CKD is not as straightforward as it is in adults. Normal hemoglobin levels vary by age, sex, and race in both adults and children [5,24,25]. Previous versions of the Kidney Disease Outcomes Quality Initiative (KDOQI) anemia guidelines (2000) simply applied the adult hemoglobin cutoff value (<11.0 g/dL) to children, which was eventually found to significantly underestimate the prevalence of anemia in patients with CKD under 18 years old [17,26,27]. Hence, such guidelines were updated in 2006 to reflect age-dependent criteria for anemia in CKD [5]. These most recent KDOQI guidelines cite reference values for children according to the data from the National Health and Nutrition Examination Survey III. They recommend diagnosing anemia when hemoglobin levels fall below the 5th percentile based on age- and sex-specific criteria and initiating appropriate evaluation for anemia. The Kidney Disease: Improving Global Outcomes (KDIGO) guidelines also based the diagnostic criteria for CKD-related anemia on age, as shown in Table 2 [28].
Treatment of anemia in CKD
The treatment of anemia is a pivotal factor in reducing morbidity and mortality rates in children with CKD and enhancing patients’ quality of life. Although the recommended frequency for assessing anemia according to KDIGO and KDOQI guidelines may vary slightly according to the CKD stage and dialysis status, generally, for primary anemia prevention in pediatric CKD, clinicians are advised to monitor hemoglobin levels at least every 6 months to 1 year for anemia detection, and if anemia is diagnosed, the hemoglobin level should be monitored at least every 1 to 3 months [5,28]. For effective anemia treatment, the two most vital components, namely, exogenous erythropoiesis-stimulating agent (ESA) and iron supplementation, must be simultaneously administered. Adjunctive therapies to supplement other nutritional deficiencies and regulate bone mineral metabolism disorders can also be combined.
ESA therapy
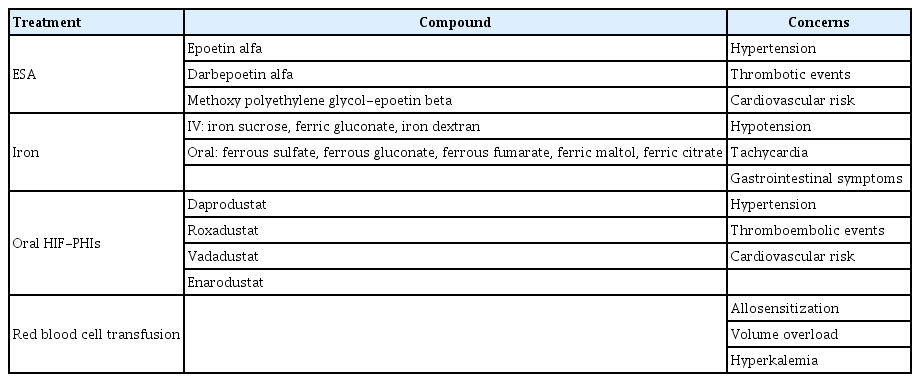
Anemia management has dramatically transformed since recombinant human EPO was introduced in the late 1980s. The ESA therapy can significantly improve anemia symptoms in patients with CKD, reducing the need for blood transfusions [29,30]. After the U.S. Food and Drug Administration (FDA) approved the recombinant human EPO (epoetin alfa) in 1989, epoetin alfa became the standard treatment for EPO deficiency anemia. Subsequently, darbepoetin alfa was approved, offering the advantage of 2 to 3 times longer half-life than epoetin alfa [31]. Additionally, methoxy polyethylene glycol-epoetin beta, which has an even longer half-life, has been used [32]. The KDIGO guidelines recommend initiating ESA therapy in adult nondialysis patients when hemoglobin is less than 10 g/dL and in adult dialysis patients when hemoglobin is 9–10 g/dL. However, no specific criteria are currently available for children with CKD, and making treatment decisions based on the potential benefits and side effects in each individual case is recommended. Furthermore, once ESA therapy is initiated, the recommended target hemoglobin concentration for maintenance therapy in pediatric patients is approximately 11–12 g/dL [28]. After the initiation, hemoglobin concentration should be measured weekly until it stabilizes and reaches the target level. The most appropriate goal during the first month of treatment is an increase in hemoglobin concentration of approximately 1 g/dL. An increase of more than 1 g/dL during the first 2 weeks of monitoring should be considered as excessive, and the ESA dose should be reduced by approximately 25% to 50% [28].
Risk of ESA treatment
One of the most recognized side effects of ESA is hypertension, which is of particular significance because of its role as both a cardiovascular risk factor and a mediator of kidney disease progression [30,33]. However, given the ease of managing hypertension through ESA dose adjustments, discontinuation of the medication is rarely necessary. The more serious side effects are related to the increased risk for thrombotic complications and cardiovascular events [34,35]. Rapid increase in hemoglobin concentration must be avoided to reduce such thrombotic risks; additionally, particular caution should be exercised in patients who have recently experienced thrombotic events or have coagulation disorders [36,37].
Target hemoglobin concentration of ESA treatment
The biggest issue arising since the introduction of ESA therapy was the standard for determining the target hemoglobin concentration [38]. Partial correction of severe anemia versus complete normalization of hemoglobin concentration through ESA therapy and their impact on the quality of life have been investigated for quite some time [39]. Most of the studies demonstrated that achieving complete normalization of hemoglobin concentration did not further improve the quality of life. Instead, more importantly, the risk for cardiovascular and thrombotic events increased [38,40,41]. In addition, ESA therapy that aims to completely normalize the hemoglobin concentration has been associated with an increased risk for complications rather than therapeutic benefits. The reason why cardiovascular complication risk increases upon the complete normalization of hemoglobin concentration remains unclear, but researchers hypothesize that this phenomenon can be explained by the increase in blood viscosity at higher hemoglobin concentrations and the increase of vascular endothelial wall stress [35]. This risk may not be caused by the elevation of hemoglobin concentration per se but rather the very high ESA dose required to normalize such concentration. Therefore, the FDA recommends maintaining ESA treatment to achieve a therapeutic effect by using the lowest ESA dose needed.
Hyporesponsiveness of ESA treatment
Although uncommon, approximately 5% to 10% of patients treated with ESA experience ESA hyporesponsiveness intermittently or chronically during ESA therapy [42]. Maintaining a hemoglobin concentration above 10 g/dL without the use of high ESA doses may be difficult for these patients. Hence, the cause of hyporesponsiveness must be investigated. Its common causes include iron deficiency, blood loss, inflammation, infection, hyperparathyroidism, and other hematologic disorders [17,42,43]. Identifying and correcting these factors first is important.
Iron therapy
The most commonly identified reason for poor response to ESA therapy in pediatric CKD with anemia is iron deficiency [44]. ESA therapy itself increases the demand for iron and may reveal or exacerbate decreased iron availability [45]. Similar to many other studies involving adult patients, supplementing iron in children undergoing ESA therapy can reduce the required ESA dose per the achieved unit of hemoglobin level [46,47]. Hence, patients with CKD must be periodically evaluated for iron status and maintained with appropriate iron agents if necessary. The goal of iron therapy is not only to replenish depleted iron but also to prevent iron store depletion, avoid iron-deficiency erythropoiesis, and maintain the target hemoglobin level. The KDIGO anemia guidelines recommend assessing the iron status at least every 3 months during ESA therapy and targeting serum ferritin levels at 100 ng/mL or more and transferrin saturation at 20% or more for the maintenance of iron therapy [28]. Iron therapy is administered either orally or intravenously, depending on the dosage and dialysis status of children with CKD [48]. Children in the nondialysis or peritoneal dialysis CKD group mostly receive oral iron agents, whereas those undergoing hemodialysis are more likely to require intravenous formulations to allow sufficient iron stores for ongoing erythropoiesis [49]. For oral iron agents, the recommended dose of elemental iron ranges from 2–3 mg/kg/day to 6 mg/kg/day, with a maximum daily dose of 150–300 mg, divided into 2–3 doses [50,51]. To maximize gastrointestinal absorption, children are advised to take iron supplements on an empty stomach and to avoid concomitant intake with any calcium-containing binding agents [52]. Although oral iron supplements are cost effective and easy to administer, their efficacy in maintaining adequate iron stores for erythropoiesis is limited because of poor gastrointestinal absorption and poor compliance. Furthermore, oral iron supplementation often falls short in keeping up with the chronic blood loss associated with chronic hemodialysis in pediatric patients [17]. According to studies, intravenous iron supplementation showed significantly better responses in terms of serum ferritin and hemoglobin level increase, iron storage, and reduced required ESA doses than oral iron supplementation [47,49]. Studies verifying the efficacy of intravenous iron in children with nonhemodialysis CKD have also been reported [53,54]. Despite the advantages of intravenous iron administration, concerns remain because of its potential association with various clinical side effects, such as hypotension, tachycardia, and gastrointestinal symptoms, in addition to inducing iron overload–related oxidative stress [55,56]. Therefore, adequate monitoring of dosages, frequency, effectiveness, and safety is essential.
Oral HIF-PHIs
Considering the risk of developing complications such as hypertension and cardiovascular events, and other adverse events associated with existing ESA medications, experiments with new drugs aimed at reducing these complications have been conducted. A new class agent of anemia treatment for patients with CKD is the hypoxia-inducible factor prolyl hydroxylase inhibitors (HIF-PHIs) [57-59]. The HIF-prolyl hydroxylase domain (PHD) pathway regulates the cellular response to hypoxia, which is also involved in anemia. HIF is transcription factor that regulates the production of EPO and is decomposed by prolyl hydroxylase under normal oxygen saturation conditions. But when oxygen is insufficient, the hydroxylation reaction cannot occur and HIF is stabilized. The HIF-PHD axis coordinates hypoxia response across multiple cell types and tissues. In addition to kidney and liver EPO production, HIF regulates iron metabolism and promotes erythrocyte precursor cell maturation and proliferation in bone marrow. HIF-PHI not only activated endogenous EPO production by inhibiting HIF-PHD, but also increases iron reuse in the liver by inhibiting hepcidin, and promotes erythropoiesis by enhancing iron availability [57-59]. Randomized, double-blind, phase 3 trial studies were conducted in adult dialysis participants with CKD-related anemia to evaluate the efficacy, safety, and pharmacokinetics between HIF-PHI and ESA therapy; results showed that the HIF-PHI agent was not inferior to ESAs in terms of correcting anemia and cardiovascular outcomes [60,61]. Based on these results, the HIF-PHI agents are currently being administered with FDA approval in adult patients undergoing dialysis for CKD, and phase 3 trial studies are underway for patients with CKD not undergoing dialysis [62,63]. Studies on the efficacy and safety of HIF-PHI, particularly daprodustat, in pediatric patients with CKD are also currently underway [64]. Unlike ESAs, which are injectable only, HIF-PHI agents are oral medications. When ESA treatment is actually used for a long period of time, most children complain of stress because the injection is too painful for them, thereby worsening their quality of life. The development of new oral agents will play a crucial role in relieving children’s stress about administering injections.
Red blood cell transfusion
Red blood cell transfusions should be used cautiously in patients with CKD, especially in children who are mostly considering future kidney transplantation, to minimize the development of allosensitization, which may affect transplant outcomes [28]. Despite the use of ESA and iron therapies, red cell transfusion is sometimes unavoidable when acute bleeding or severe anemia symptoms occur. Red blood cell transfusion can provide rapid improvement in symptoms related to anemia clinically, but in patients with CKD, particular attention must be paid to various risks, such as volume overload, hyperkalemia, citrate toxicity, hypothermia, coagulopathy, and immunologic reactions [65]. Therefore, red blood cell transfusion is a treatment option for patients with chronic anemia whose ESA treatment is not sufficiently effective due to conditions such as hemoglobinopathy, bone marrow failure, and ESA resistance. It may also be considered in patients who require rapid hemoglobin correction preoperatively [28]. As in adults, red blood cell transfusion, along with immunosuppressant administration, before kidney transplantation may lead to a better transplant outcome among children [66]. Finally, treatment of anemia in children with CKD was summarized in Table 3.
Conclusion
Anemia is an important complication associated with several adverse outcomes in children with CKD. EPO deficiency and iron deficiency act as the most important etiological factors of anemia; thus, ESA therapy with iron supplementation is the best treatment for CKD-related anemia. Providing the appropriate treatment for this type of anemia is crucial because it can not only minimize discomfort in daily life (e.g., school attendance) by improving the quality of life, cognitive function, and cardiovascular function but also reduce hospitalization risk, rapid aggravation of kidney failure, and mortality.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Author contributions
All the work was done by MJP and MHC.