Rapid Resolution of Atypical Hemolytic Uremic Syndrome by Eculizumab Treatment
Article information
Abstract
Atypical hemolytic uremic syndrome (aHUS) is an extremely rare and life-threatening disorder. Typical HUS is often caused by Shiga toxin-positive Escherichia coli, while aHUS is caused by dysregulation of the alternative pathway of the complement system in association with genetic abnormalities or development of autoantibodies. Eculizumab, a humanized anti-complement 5 monoclonal antibody, is recommended for the treatment of aHUS, but its long-term safety and efficacy in pediatric patients remain under review. In this paper, we report a pediatric case of aHUS with anti-complement factor H autoantibodies, who was treated successfully with eculizumab.
Introduction
Thrombotic microangiopathy (TMA) is characterized by microangiopathic hemolytic anemia, thrombocytopenia, and vital organ damage, most frequently in, but not limited to, the kidney [1,2]. In children, a type of TMA, hemolytic uremic syndrome (HUS), is relatively common and is an important cause of childhood acute kidney injury. Most cases of childhood HUS are typical of hemorrhagic diarrhea, which is caused by Shiga toxin-producing Escherichia coli (STEC) [3]. Meanwhile, atypical HUS (aHUS) is extremely rare, more severe than STEC-HUS, and often life-threatening. Recently, aHUS was revealed to be caused by dysregulation of the alternative pathway of complement activation [4-6].
Uncontrolled complement activation is often due to genetic abnormalities in components of the alternative pathway, most commonly complement factor H (CFH), the most important regulatory protein, or autoantibodies against regulatory proteins, commonly CFH (anti-CFH autoantibodies). Such uncontrolled complement activation leads to uncontrolled cleavage of the terminal complement protein complement 5 and excessive complement 5b-9 complex (membrane attack complex [MAC]) formation, resulting in endothelial cell injury and activation leading to TMA [5-7].
Eculizumab, the first recombinant humanized monoclonal anti-complement 5 antibody, inhibits cleavage of complement 5, thereby preventing MAC formation [8]. This medication was first approved as a treatment for paroxysmal nocturnal hematuria, where red blood cells are destroyed by MAC. Then, it was proven to be effective in aHUS and was approved for this indication in Korea in 2018. Nowadays, eculizumab is regarded as the first-line treatment for aHUS [5-7].
In this paper, we report a case of aHUS in a child successfully treated with eculizumab.
Case report
A previously healthy 7-year-old girl was transferred to the emergency department of our hospital from an outside hospital due to general weakness, dyspnea, and pallor. She had mild cough and rhinorrhea. A few days ago, she had one episode of non-projectile/non-bilious vomiting, and urinated dark-blood colored for a day. She did not have fever, diarrhea, or abdominal pain. Upon presentation, her blood pressure (BP) was 128/61 mmHg (>99th percentile of systolic BP, 67th percentile of diastolic BP), pulse rate was 111 beats/min, respiratory rate was 28 breaths/min, and oxygen saturation in room air was 100%. She was alert, pale-looking, and had no other specific signs, such as definite oliguria or body weight gain, except hypertension. Her abdomen was soft without tenderness or rebound tenderness. Initial laboratory findings were as follows: hemoglobin (Hb) was 4.9 g/dL, platelet was 48,000/uL, lactate dehydrogenase was 2,511 IU/L, plasma hemoglobin was 39.2 mg/dL, reticulocyte count was 5.61%, haptoglobin was <7 mg/dL, serum total bilirubin was 1.5 mg/dL, blood urea nitrogen was 134 mg/dL, creatinine level was 3.58 mg/dL, eGFR was 13.84 mL/min/1.73 m2, complement 3 level was 52 mg/dL, and complement 4 level was 32 mg/dL. A peripheral blood smear revealed schistocytes (8.5/high power field). Hematuria (30–49 urine red blood cell/high power field) and proteinuria (7.38 mg/mg urine protein/creatinine ratio) were also found. Abdominal ultrasonography revealed normal morphology of the kidneys, and renal Doppler ultrasound showed slightly increased renal cortical echogenicity, diminished renal perfusion, and markedly elevated resistive index value of around 1.0.
Because of severe uremia, she started hemodialysis on the first day of hospitalization and daily infusion of fresh frozen plasma. Since the patient had no history of diarrhea, aHUS was suspected. After work-up for the differential diagnosis of TMA, plasma was infused along with daily emergency hemodialysis. However, azotemia and hemolysis persisted, and plasmapheresis was performed on the 3rd day to provide complement alternative pathway regulators and remove probable inhibitors of regulators, since eculizumab, the first-line treatment of aHUS, was not available until submission of documents for approval of insurance coverage of this medication from the Health Insurance Review & Assessment Service. Before plasmapheresis, blood samples were collected, and the ADAMTS-13 activity test was performed to discriminate TTP (thrombotic thrombocytopenic purpura). In addition, a Shiga toxin polymerase chain reaction (PCR) assay and stool culture were performed to discriminate STEC-HUS. TTP and STEC-HUS could be excluded through the normal range of ADAMTS-13 activity of 96% (normal range 44–121%) and negative results of stool PCR and culture. Upon submission of the necessary documents and confirmation of the diagnosis of aHUS, eculizumab was administered on the 4th day after presentation. It was started with a dose of 600 mg weekly for 2 doses and then maintained at a dose of 600 mg every 2 weeks.
Before eculizumab treatment, meningococcal vaccination and prophylactic antibiotics for Neisseria meningitidis infection were given. These prophylactic antibiotics against invasive meningococcal infection were administered to cover the first 2 weeks after vaccination until the formation of acquired immunity. Initially, rifampin was started, but a rash occurred as an adverse effect, prompting a change to ciprofloxacin instead.
With the administration of eculizumab, her hemogram started to recover rapidly and her platelet count normalized after 10 days, followed by normalization of Hb levels. Her renal function started to recover 7 days after eculizumab therapy, and normalized 27 days later (Fig. 1). At eleven months, the patient was still managed with biweekly eculizumab therapy with no recurrence of aHUS and had her kidney function totally restored without renal replacement therapy.
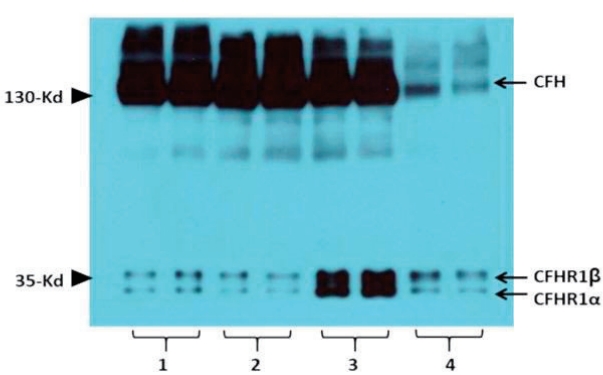
Several laboratory tests to identify underlying abnormalities in the complement system were performed. Western blot analysis revealed a normal concentration of CFH with increase concentration of CFHR1 in the patient’s plasma (Fig. 2). Western blot analysis using a commercial polyclonal anti-human complement factor H (CFH) antibody shows three bands in the plasma of normal controls (lanes 1 and 2): upper thick band of CFH, lower two bands of CFH-related protein 1α (CFHR1α) and 1β (CFHR1β). While the concentration of CFH of the patient was normal, lower two bands were thicker than others, which suggests increased concentration of CFHR1 protein (lane 3). Lane 4 was a patient with atypical hemolytic uremic syndrome, biallelic mutations in CFH, and near total deficiency of plasma CFH protein.
However, the multiplex ligation-dependent probe amplification (MLPA) study to document copy number variations in CFHRs was not performed. A panel gene sequencing for TMA covering 15 candidate genes revealed no pathogenic mutation. A test for plasma anti-CFH immunoglobulin G (IgG) using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (CFH IgG ELISA kit; Abnova, Heidelberg, Germany) revealed a markedly higher titer than in normal subjects (Table 1).
For the management of anti-CFH antibodies, we started immunosuppressive treatment with steroids, followed by mycophenolate mofetil (MMF). At the last follow-up, 11 months after presentation and 5 months on immunosuppression, anti-CFH antibodies remained positive, and eculizumab treatment was continued, according to the protocol.
Discussion
Before the introduction of eculizumab, plasma therapy (plasma exchange or plasma infusion) have been the standard treatment of aHUS. But plasma therapy was not effective; historically, aHUS had a poor prognosis with significant morbidity and mortality, with up to 50% of patients progressing to end-stage renal disease (ESRD), and 10 to 15% mortality rate during the acute phase. Treatment with eculizumab was proven to be effective in aHUS, substantially improving patient survival and preventing progression to ESRD and disease recurrence [6,9,10].
Eculizumab in aHUS led to hematological normalization and reduced renal injury with significantly improved kidney function. Because earlier initiation with eculizumab is associated with the best reversibility of hematological and renal recovery with discontinuation of hemodialysis, treatment with eculizumab should be started as early as possible after the diagnosis of aHUS [3-6]. In our patient, eculizumab was started on the 4th day and was still treated with eculizumab without relapses or disease activity in the long term.
Although eculizumab is proven to be effective as an initial treatment for aHUS because it is a complement inhibitor, it increases life-threatening infections by encapsulated microorganisms, such as Neisseria meningitidis, Streptococcus pneumoniae, and Haemophilus influenzae[3,5,8,9]. Therefore, patients treated with eculizumab should be vaccinated against these organisms at least 2 weeks prior to initiating treatment. If the patient could not be vaccinated before eculizumab treatment, appropriate antibiotics should be administered for 2 weeks for prophylaxis against bacterial meningitis [5,7].
aHUS results from genetic mutations of CFH or anti-CFH autoantibodies. There are studies that identified an association between anti-CFH autoantibodies and CFHR1 deletion in aHUS. Anti-CFH autoantibodies are considered to interfere the regulatory function of CFH on alternative pathway of complement activation, probably by interfering the binding of CFH to C3. Studies have shown that CFHR1 deletion is associated with an increased risk of aHUS [11]. In cases of CFHR deletion, a western blot of CFH often reveals a lack or decrease in CFHR due to cross-reactivity between CFH and CFHRs. Therefore, a western blot of CFH can screen patients with CFHR deletions if the respective mutation results in a decrease or lack of CFHRs. However, in our patient, a western blot of CFH revealed a thicker band of CFHR. We suspect that this implies the multiplication of CFHR genes, but a gene panel analysis of relevant genes did not detect any mutation in CFHRs. Since copy number variation is not detectable through targeted gene sequencing, and CFH and CFHRs are notorious for evading detection of variation due to their homology, the MLPA of CFHR is necessary to accurately detect dose variations in these genes. However, this is not clinically available; therefore, we could not pursue this relevant investigation.
According to Loirat et al., in anti-CFH antibody-related aHUS, initial treatment is eculizumab within 24 to 48 hours after onset. After positive and anti-CFH antibodies are confirmed, they suggest adding corticosteroids and/or mycophenolate mofetil (MMF) or azathioprine while maintaining eculizumab. Although eculizumab is proven to be an effective initial treatment for aHUS, it is not supposed to be effective in inhibiting anti-CFH antibody production [12,13] . Accordingly, we started corticosteroids and MMF after detecting anti-CFH antibodies, although our patient continued to perform well with biweekly eculizumab therapy with no recurrence. The patient still had a significant amount of anti-CFH antibodies at the last follow-up. Once the anti-CFH autoantibody disappears, eculizumab can be discontinued while continuing immunosuppressants.
In conclusion, this case shows the efficacy of eculizumab in promptly reversing aHUS in a patient with anti-CFH autoantibodies, and in maintaining adequate renal function for more than 12 months without recurrence.
Acknowledgements
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI18C0013).
We would like to thank Editage (www.editage.co.kr) for English language editing.
Notes
Patient consent
This study was approved by the institutional review board (IRB), and the consent was waived due to the nature of the retrospective study [IRB number 2004-199-1119].
Conflict of interest
No potential conflict of interest relevant to this article was reported.