An Epidemiologic Study on Hosts and Pathogens of Urinary Tract Infection in Urban Children of Korea (2012–2017)
Article information
Abstract
Purpose
We aimed to determine characteristics of host, causative organisms, and antibiotic susceptibility of bacteria in pediatric patients with UTI living in metropolitan area of Korea.
Methods
Retrospective investigation was done for the causative organisms of UTI in 683 pediatric cases treated at Ajou University Hospital from 2012 to 2017. Patients were classified into Escherichia coli and non-E. coli group, where E. coli group was subdivided into ESBL(+) and ESBL(-) groups based on whether the bacteria could produce extended spectrum beta-lactamase (ESBL). Antibiotic susceptibility of the causative organism was also determined.
Results
A total of 683 UTIs occurred in 550 patients, of which 463 (67.8%) were first-time infection and 87 (32.2%) were recurrent ones (2–7 recurrences, 2.52 average), and 64.9% were male and 35.1% were female. The most common causative organism was E. coli (77.2%) and ESBL(+) E. coli was found in 126 cases. The susceptibility of E. coli to 3rd or 4th generation cephalosporin was relatively higher than that to ampicillin or amoxicillin/clavulanic acid. ESBL(+) E. coli showed higher resistance rate to 3rd or 4th generation cephalosporin than ESBL(-) E. coli.
Conclusion
New treatment guideline should be considered due to the incidence of ESBL(+) E. coli increased up to one quarter of UTI cases.
Introduction
Urinary tract infection (UTI) is one of the most common bacterial infections in children. It is the most common cause of fever without focus in infants younger than two years of age. It accounts for 5–14% of pediatric emergency room visits [1,2]. The prognosis of UTI is good in general. However, over 60% of children with febrile UTI might experience ongoing renal damages resulting in early hypertension, renal insufficiency, or end stage renal failure in adulthood [3,4]. To prevent renal loss from UTI, it is essential to know the pathophysiology of the disease, including host factors, bacterial factors, and antibiotic resistance [5,6]. The objective of this study is to determine the recent characteristics of host, pathogens, and antibiotic susceptibility of bacteria in pediatric patients with UTI living in metropolitan area of Korea for successful treatment.
Materials and methods
A total 683 pediatric patients aged below 15 years treated for UTI from January 2012 to December 2017 were included in this study. Patients were divided into five age groups (<6 months, 6–12 months, 12–24 months, 24–60, and ≥60 months). Pathogenic strains were grouped as E. coli or non-E. coli according to Gram staining, and E. coli group was subdivided into ESBL(+) and ESBL(-) groups depending on whether the bacteria could produce extended spectrum beta-lactamase (ESBL).
UTI was confirmed by body temperature greater than 38℃ using a tympanic thermometer, urine leukocytes of more than 10 per high power field view on microscopy, and colony count of pathogen at more than 105 colonies/mL of single organism on urine culture study. When two or more strains were cultured, the cases were excluded from this study [7-9].
Urine was collected in a sterilized urine bag or plastic cup after cleaning the urethral orifice area with 2% boric acid. For microscopic examination, urine samples were centrifuged and Gram stained to observe urinary bacteria and leukocytes. For culture study, 0.001 mL urine was inoculated into blood agar medium and MacConkey agar medium separately and incubated at 37℃ for 18–24 hours. The number of bacterial colonies per 1 mL was then calculated [10]. Minimal inhibitory concentration (MIC) to the strain was determined using VITEK2 (Bio-Merieux), a microbial automation device according to the Clinical and Labora tory Standards Institute (CLSI). Bacteria, more than 105 colonies/mL were considered to be significant as causative agents of UTI.
The antibiotic resistance of E. coli strains was analyzed. Resistance rate was expressed as {(Intermediate+Resistance)/(Susceptible+Intermediate+Resistance)} for each susceptible, intermediate, and resistant strain to each antibiotic. Bacterial strains with moderate susceptibility in antibiotic sensitivity tests were classified as resistance.
Collected data were analyzed using SPSS 18.0 program. Differences according to gender and age groups were evaluated using Chi-square and t tests. In order to evaluate the difference in incidences among the age groups, goodness of fit was determined by Chi-square test, and cross-tabulation analysis was done to establish significant differences in the incidence of UTI, by gender among the age groups. Statistical significance was considered when P value was less than 0.05.
Results
1. Age and gender distribution of UTI patients
Among 683 cases, 443 (64.9%) were male and 240 (35.1%) were female. Patients’ age less than 6 months accounted for the majority (n=335, 49%), followed by 6–12 months (n=192, 28%), 12–24 months (n=53, 7.8%), 24–60 months (n=50, 7.4%) and ≥60 months (n=53, 7.8%). Thus, 77.2% of childhood UTI developed at the age under 24 months. The incidence of UTI in age <6 months group was significantly higher than others (P <0.001), and female incidence was higher only in 24–60 months age group compared to other female age groups (P<0.001, Table 1).
2. Identification of etiologic strains of UTI
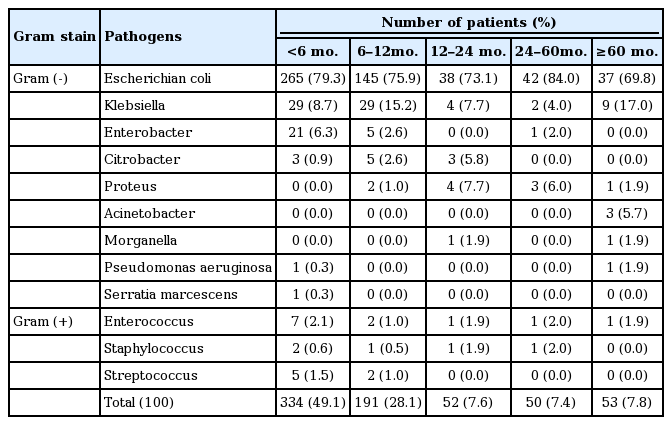
Escherichia coli occurred in most cases (n=527, 77.2%), followed by Klebsiella species (n=73, 10.7%) and Entero bacter species (n=27, 4.0%). Among 24 patients who showed unusual pathogenic strains of UTI such as Staphylococcus, Enterococcus, and Streptococcus, 10 (42%) were found to have underlying disease or conditions such as De Lange syndrome, cerebral palsy, corpus callosum agenesis, cerebral infarction, ridden state, and systemic steroid use (Table 2).
3. Distribution of pathogens according to the age of patients
Escherichia coli was the most common etiologic strain in all age groups (69.8% to 79.3%), and Klebsiella species was the second most common (4.0% to 17.0%). Citrobacter was identified 0.9–2.8% in under 24 months age groups and, Enterobacter 2.6–6.3% in under 12 months. Proteus mirabilis showed low frequency (1.0–7.7%) after 6 months of age (Table 3).
4. Incidence of Escherichia coli according to age and gender of patients
Incidence of Escherichia coli as the etiology of UTI was the highest (at 84.0%) in the age group of 24–60 months. It was not significantly different by gender (77.0% in males vs. 77.5% in females, P =0.876). There was no significant difference among age groups either (P =0.302) (Table 4).
5. Incidence of ESBL(+) Escherichia coli by age and gender of patients
Among a total of 527 E. coli -identified cases, ESBL-producing, ESBL(+) E. coli strains were found in 126 (24%) cases, including 89 (70.6%) males and 37 (29.4%) females. Frequencies of ESBL(+) E. coli were not significantly different according to gender or age of patients (P =0.110 or P =0.100, respectively) (Table 5).
6. Annual incidence of ESBL(+) Escherichia coli
A total of 126 ESBL(+) E. coli cases were found, including 5 (4.0%) in 2012, 22 (17.5%) in 2013, 12 (9.5%) in 2014, 16 (12.7%) in 2015, 25 (19.8%) in 2016, and 46 (36.5%) in 2017. There was an obvious incremental tendency in the occurrence of ESBL(+) E. coli cases by year (Fig. 1).
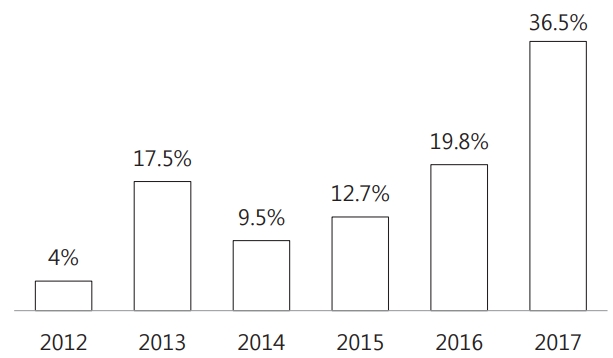
Annual number of patients with ESBL-producing Escherichia coli in children with UTI living in urban areas of Korea (2012–2017). Extended spectrum beta-lactamase producing, ESBL(+) E.coli strains were found in 126 patients during the last six years, and the annual report of its occurrence gradually increased from 5 (4.0%) in 2012, to 46 (36.5%) in 2017. However, there was no statistical significance in the incidence of ESBL(+) E. coli among total urinary tract infections (P=0.097).
Over a six year-period from January 2012 to December 2017, the occurrence of ESBL(+) E.coli case has increased from 25.0% in 2012 to 31.9% in 2017. However, its incidence was not significantly different during these years (P =0.097, Table 6)
7. Antibiotic resistance of general and ESBL(+) strains of Escherichia coli
As a result, the resistance rate of ampicillin was 71.7% and that of amoxicillin/clavulanic acid was 33.6%. Resistance rates against third- and fourth- generation cephalosporin were 23–26%. Resistance rate to ertapenem, imipenem, and tigecycline was 0.0%. Antibiotic resistance rates of ESBL(+) E. coli to ampicillin, amoxicillin/clavulanic acid, and third/fourth generation cephalosporin were significantly high; 100.0%, 49.2%, and 90.0%, respectively. Their resistance rate to ertapenem, imipenem, and tigecycline was 0.0%, which was the same as the resistance rate of all E. coli strains. Resistance rates to antibiotics other than amikacin were statistically significant between ESBL(+) and ESBL(-) E. coli (P<0.05, Table 7).
8. Incidence of ESBL(+) pathogens according to recurrence rate of UTI
Among 550 patients, ESBL(+) was identified in 146 (26.5%). Among these, 126 were E. coli and 20 were Klebsiella species. UTI recurred 220 times in 87 patients; averaging 2.52 and up to 7 times per patient. The recurrence rates of UTI seems to be related with the incidence of ESBL(+), but there was no significant difference between the frequency of ESBL(+) and relapses of UTI (P = 0.728, Table 8).
9. Incidence of VUR according to UTI recurrences
No significant statistical differences were found in the distribution of the severity of vesico-ureteral reflux (VUR) between recurrent (n=87) and non-recurrent UTI groups (n=463) (P =0.194). However, VUR was present significantly higher in recurrent group (45.9%, 34 out of 74 patients) than in non-recurrent group (33.5%, 77 out of 230 patients) (P =0.002, Table 9). In 87 recurrent UTI patients, the rate of ESBL(+) was not different by the degree of VUR (P =0.477), nor to the presence of VUR (P =0.618, Table 10).
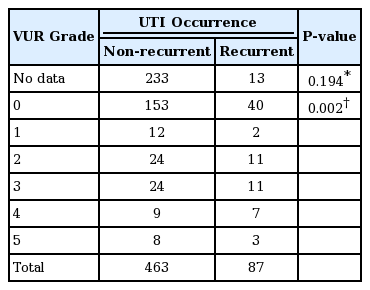
Incidence and Severity of VUR according to the Recurrence of Infections in Children with UTI (2012–2017)
Discussion
Among the 683 pediatric cases diagnosed with UTI at our institute from 2012 to 2017, 64.9% were male and 35.1% were female. This is similar to previous studies showing a higher prevalence in male children including neonatal period, and higher incidence in females after childhood, except for those aged above 50 years [11]. High incidence rate of UTI in lower age groups of male might be due to poor penile hygiene during diaper wearing period. Current study indicated the prevalence of UTI in females was lower than that of males in early childhood, but then increased with age: from 24.2% in age <6 months, to 58.2% in age >5 years (Table 1).
Several investigators have reported that pathogens of UTI are mainly enterobacteria, including E. coli and Enterobacteriaceae. Hooton and Stamm have reported that in the United States, E. coli , Staphylococcus, Enterococcus, and other Gram-negative organisms account for 75-90%, 5–15%, and 5–10% of all UTI, respectively [12]. In the present study, the ratio of E. coli was highest at 77.2%, followed by Klebsiella at 10.7% and Enterobacter at 4.0%, showing that the portion of Gram-negative bacterial infections other than E. coli should be taken into account [13,14]. UTI is mostly caused by ascending route through colonization and propagation of pathogens of lower gastrointestinal tract to perineum, urethral opening, inside urethra, then finally to the kidney [1]. Consequentially, most common causative organism of UTI is E. coli , while non-E. coli cases account for about 10-35% [15-17]. We have a similar result of non-E. coli UTI at 23%, whilst a domestic study reported 32.9% [18].
We investigated the frequency of ESBL-producing ESBL(+) E. coli strains. UTI caused by ESBL(+) E. coli has been increasing in numbers for the last 6 years. The average prevalence rate of ESBL(+) E. coli was 24.0% in this study, which is lower than those of foreign reports, at 33–69%. However, the incidence has increased up to 36.5% in 2017, drawing more attention for ESBL(+) E.coli infections. It was also found that 113 out of 463 patients were ESBL(+) at the time of their first visit, which led to estimate the community prevalence rate of ESBL(+) uropathogen to be around 24.4% in children (Table 8).
In this study, antibiotics with resistance rates below 10% were amikacin, cefoxitin, ertapenem, imipenem, piperacillin/tazobactam, tigecycline, and nitrofurantoin. Third- and fourth- generation cephalosporin such as cefepime (23.7%) and cefotaxime (26.9%) showed relatively low resistance rates, whereas ESBL(+) E. coli showed high resistance rate to third- and fourth-generation cephalosporin such as cefepime (99.2%) and cefotaxime (99.2%). However, a considerably low resistance rate was displayed against carbapenem series antibiotics such as ertapenem (0.0%) and imipenem (0.0%). For E. coli not producing ESBL, it seems reasonable to consider third- and fourth-generation cephalosporin as the first line antibiotics. For patients with ESBL(+) E. coli cultured or serious clinical conditions, carbapenem series might need to be considered initially.
Nationally, the common regime for UTI treatment is to give empirical antibiotics firstly right after diagnosis, and to discharge the patient or schedule the next visit, if there are apparent clinical improvements for a few days. After completing two weeks of antibiotics administration, the patient will come and receive tests for confirming successful treatment. At times, the patient is informed late that he or she had ESBL(+) bacterial infection, even though the final follow-up tests are normal: no clinical sign, urine culture negative, and no pyuria. For such cases, we should pay close attention to prevent incomplete treatment or recurrence of UTI.
Throughout the current cases, the recurrence rate of UTI between ESBL(+) and ESBL(-) patients showed no significant difference (Table 8), but greater in patients with vesicoureteral reflux (VUR). Severity of VUR did not affect the recurrence (Table 9).
In Korea, antibiotics are occasionally prescribed for treating simple pediatric febrile illnesses at primary care institutions, and this may lead to increases in antibiotic resistance of bacteria and also to more serious renal damage, due to delayed or incomplete treatment in those misdiagnosed UTI as simple pharyngitis or acute otitis media. It was acknowledged that susceptibility of E. coli to third- or fourth- generation cephalosporin was higher than that to ampicillin or amoxicillin/clavulanic acid. Also, ESBL(+) E. coli showed higher resistance rate to third- or fourthgeneration cephalosporin than ESBL(-) E. coli . As a result, empirical choice of antibiotics for UTI treatment may not be safe anymore due to increased frequency of ESBL(+) E. coil up to one quarter of the cases. Therefore, treatment guideline for UTI should be looked into, reflecting the recent findings of antibiotic resistance of pathogens and host characteristics.
Choosing antibiotics for UTI treatment will become more difficult as the incidence of ESBL(+) E.coli is becoming more common. The success rate of primary treatment with empirical antibiotics will be lowered, and, the renal damages will be more serious.
The number of research subjects was much reduced, because this study included single bacterium cultured cases exclusively. In order to obtain more accurate and useful information for UTI treatment, additional researches should be done through extension of research period and expansion of inclusion range of patients.
Notes
Patient consent
This study was approved by the institutional review board (IRB), and the consent was waived due to the nature of the retrospective study [IRB number MED-MDB-18-342].
Conflict of interest
No potential conflict of interest relevant to this article was reported.